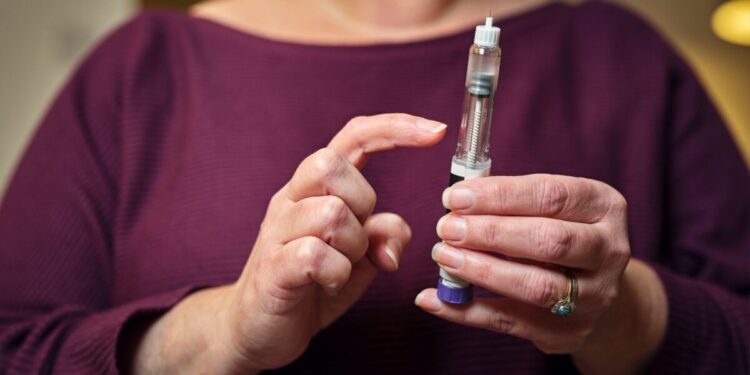
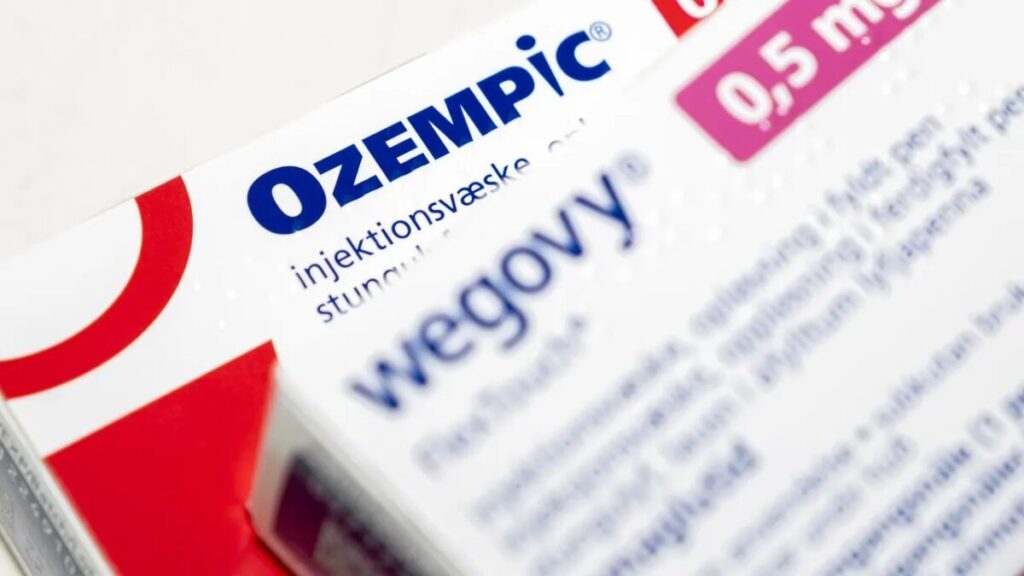
India could soon expand access to weight-loss drugs dramatically as the patent on semaglutide, the molecule behind Novo Nordisk’s blockbuster Wegovy and Ozempic, expires Friday, triggering a rush of cheaper generic versions that could slash prices by more than half and transform treatment for millions with diabetes and obesity.
Investment bank Jefferies has called it a potential “magic-pill moment” for India, predicting the semaglutide market could eventually reach $1 billion domestically with the right pricing and uptake. Analysts expect around 50 branded semaglutide generics to enter the market within months, a familiar pattern in India’s fiercely competitive pharmaceutical industry. When the diabetes drug sitagliptin went off patent in 2022, about 30 branded versions appeared within a month and nearly 100 within a year.
Several Indian drugmakers are already preparing to make the move. According to Sheetal Sapale, vice-president at research firm Pharmarack, major firms including Cipla, Sun Pharma, Dr Reddy’s Laboratories, Biocon, Natco, Zydus and Mankind Pharma are readying branded generics. Prices are expected to fall sharply. Current monthly treatment costs are steep: Ozempic typically sells for 8,800 to 11,000 rupees ($95-$119), while Wegovy can cost 10,000 to 16,000 rupees ($108-$173). Sapale expects generic competition to push that down to roughly 3,000 to 5,000 rupees ($36-54) per month.

Lower prices could transform the market. India’s anti-obesity drug sector, covering both injectables and oral medicines, has already grown rapidly, from roughly $16 million in 2021 to close to $100 million, according to Pharmarack. The surge reflects a broader health shift. India already has more than 77 million people with type-2 diabetes and one of the world’s largest populations of overweight adults.
Muffazal Lakdawala, a Mumbai-based bariatric surgeon, says these drugs could dramatically expand treatment for India’s large population of patients with diabetes and obesity. “It is great that these will become cheaper so that more of the diabetic and obese Indian population can access them,” he says. But he adds a warning: “The quality of the drugs being made here must be very tightly regulated.”
That caution reflects a broader reality about India’s pharmaceutical industry, the global powerhouse behind low-cost generic medicines. The country is the world’s largest supplier of generic medicines, producing roughly 60,000 brands across more than 60 therapeutic categories and accounting for around 20% of global generic supply. “The export potential of Indian generic weight-loss drugs is humongous,” says Namit Joshi, chairman of Pharmaceuticals Export Promotion Council of India. “The US market alone could scale to $10 billion within a few years as obesity rates drive demand.”
Yet enthusiasm among doctors remains tempered by caution. GLP-1 drugs are powerful but not risk-free. Side-effects can include nausea, vomiting and digestive problems; rarer complications include gallstones or pancreatitis. Mumbai-based diabetologist Rahul Baxi says success depends on “right patient selection” and warns patients often misunderstand the role of the drugs. “People come asking to lose 10kg in three months,” Baxi says. Another challenge is that weight loss often reverses once the drugs stop. “If you stop the drugs, the appetite comes back voraciously,” Baxi says.
There are also concerns about misuse as prices fall. Physicians report patients being prescribed high doses by gym trainers, beauty clinics or dieticians with no authority to do so. “More access to cheap drugs means a higher chance of abuse,” says Bhaumik Kamdar, a Mumbai-based chest physician. “Access calls for more responsibility and stricter regulation. I am cautiously optimistic about these drugs.”
The government is trying to temper the hype. In an advisory last week, India’s drug regulator warned pharmaceutical companies against promoting prescription weight-loss medicines such as GLP-1 drugs directly to consumers. For regulators and doctors alike, the coming months may test whether India can balance affordability with oversight. “I am actually writing on prescriptions to many patients: Come to me after 20 March when the prices come down,” says Baxi.